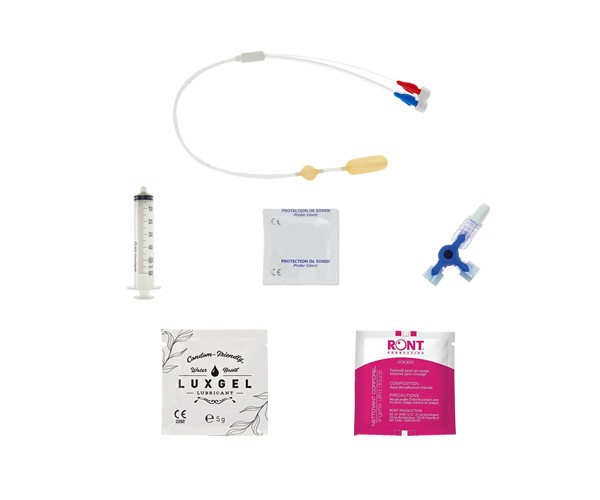
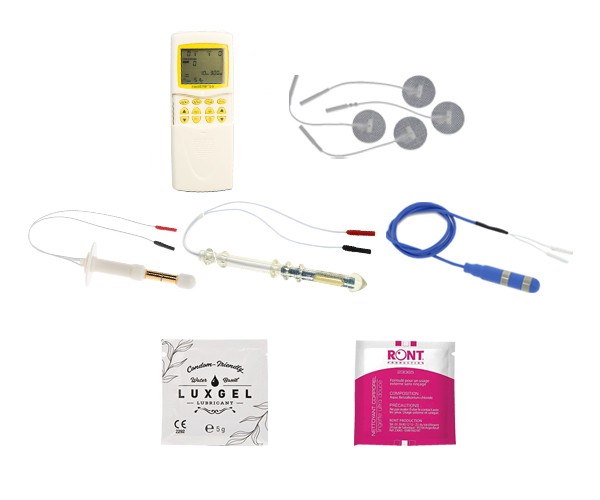
DÉCOUVREZ NOS STIMULATEURS
Découvrez nos stimulateurs portables pour pouvoir vous rééduquer chez votre thérapeute comme à domicile.
Actualités & conseils
Conseils pratiques : comment gérer et apaiser les règles douloureuses ?
Découvrez nos conseils pratiques pour surmonter les défis des règles douloureuses. Que vous recherchiez des moyens naturels pour soulager la douleur ou des...
Le plancher pelvien chez la femme
Découvrez l'importance d'un plancher pelvien fort pour la santé des femmes. Ce groupe de muscles et de tissus joue un rôle crucial dans la continence, la...
La rééducation périnéale et l'assurance maladie : quelle prise en charge pour...
La rééducation périnéale est cruciale pour la santé physique et le bien-être, mais comment votre assurance maladie la couvre t-elle ? Cet article explore la...
et fabriquées en Europe


.png)