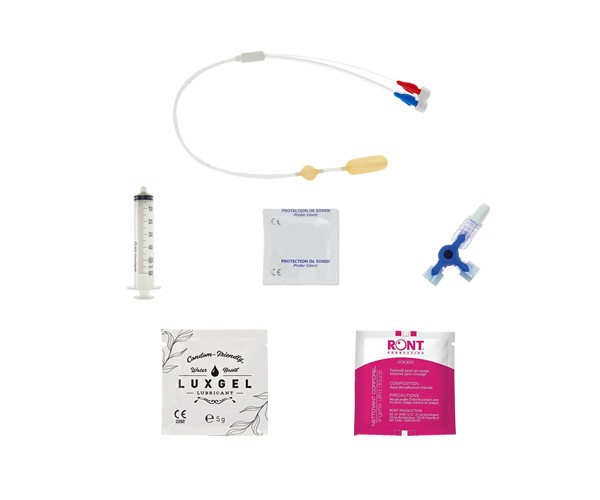
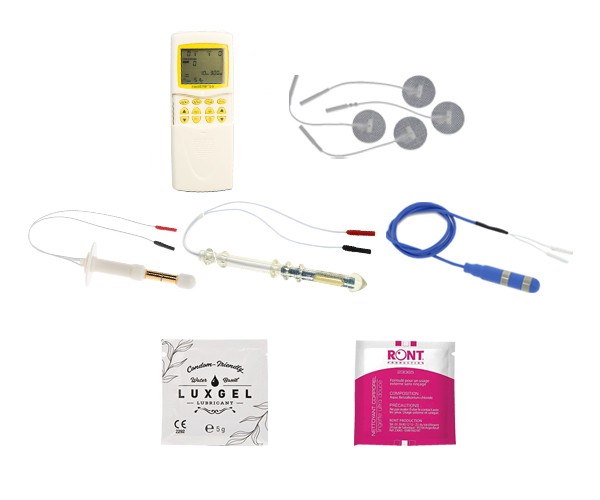
DÉCOUVREZ NOS STIMULATEURS
Découvrez nos stimulateurs portables pour pouvoir vous rééduquer chez votre thérapeute comme à domicile.
Actualités & conseils
L’endométriose : comprendre une maladie complexe et ses répercussions
Découvrez l'univers complexe de l'endométriose à travers notre dernier article. Cette maladie gynécologique chronique, souvent méconnue, affecte des millions...
Révolution dans l'hygiène féminine : tout ce que vous devez savoir sur les...
Plongez au cœur de la révolution de l'hygiène féminine avec notre guide complet sur les coupes menstruelles réutilisables ! Explorez une alternative...
Conseils pratiques : comment gérer et apaiser les règles douloureuses ?
Découvrez nos conseils pratiques pour surmonter les défis des règles douloureuses. Que vous recherchiez des moyens naturels pour soulager la douleur ou des...
et fabriquées en Europe


.png)